Understanding Trevogrumab (REGN1033)
Trevogrumab (REGN1033) is an investigational monoclonal antibody that targets myostatin, a protein that restricts muscle growth. By blocking this signal, you may preserve or increase lean muscle mass in clinical settings where muscle loss is a concern.
Researchers are studying trevogrumab in controlled trials, including combinations with GLP-1 receptor agonists for weight management. It does not have FDA approval and remains under active clinical evaluation.
How Trevogrumab Works
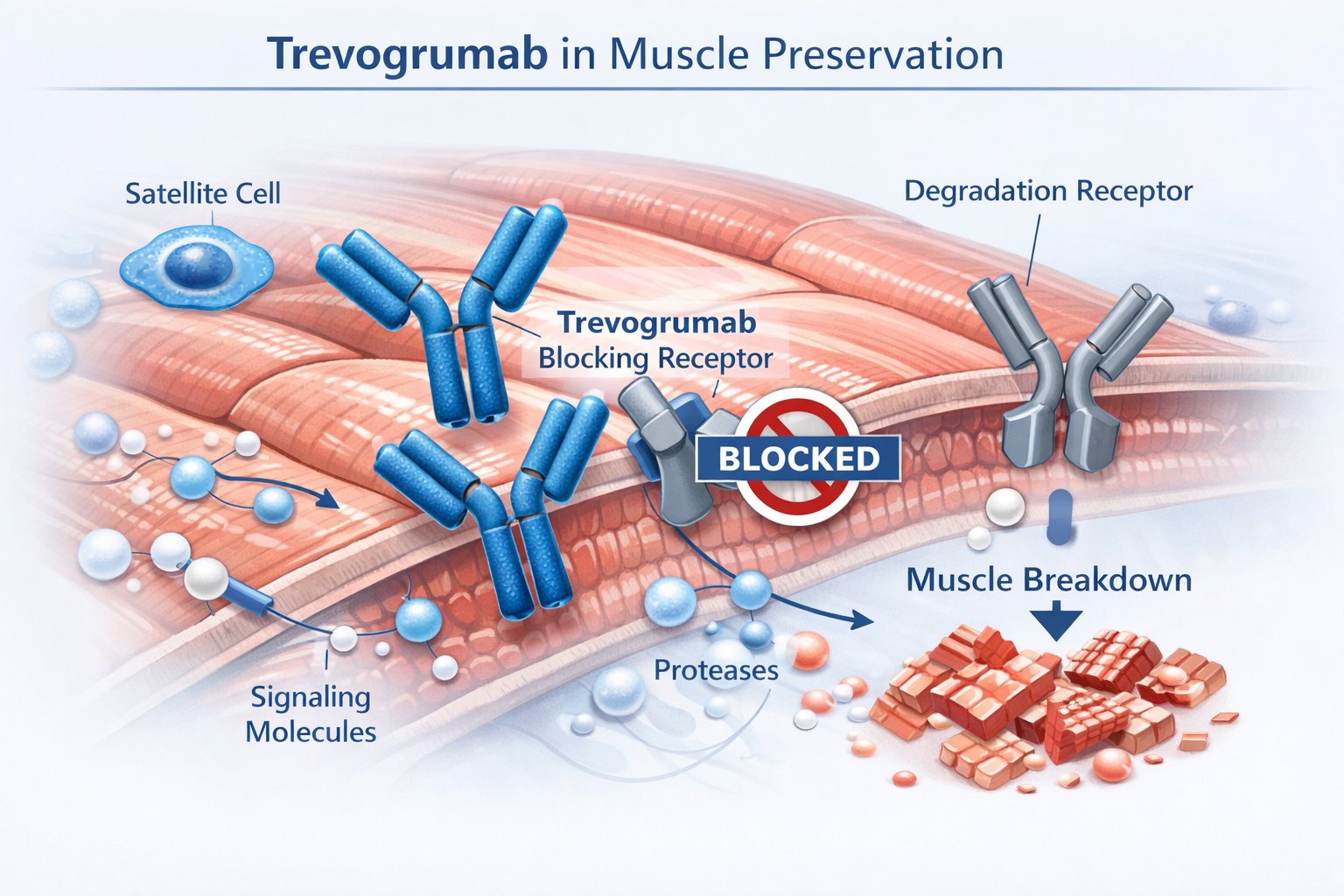
Trevogrumab acts as a human monoclonal antibody that targets myostatin (GDF8), a protein that restrains muscle growth. As an anti‑GDF8 myostatin inhibitor, it binds directly to this signaling molecule and limits its biological activity.
Understanding the Myostatin Signaling Cascade
Myostatin normally signals muscle cells to slow growth and reduce protein synthesis. When you block this pathway, you reduce its suppressive effect on skeletal muscle tissue.
By neutralizing myostatin, and potentially complementing strategies that target activin A with an anti‑activin A agent, you may support greater muscle protein production and lean mass preservation.
Trevogrumab in the Phase II COURAGE Clinical Trial
The Phase 2 COURAGE trial (NCT06299098) evaluated trevogrumab alongside semaglutide in adults with obesity. Researchers designed this clinical trial to assess changes in body composition, safety, and tolerability over 26 weeks.
In the Phase II COURAGE trial, participants who received semaglutide alone showed reductions in lean body mass during weight loss. When investigators added trevogrumab, lean mass decline appeared less pronounced.
Early reports also suggested that combining therapies shifted weight reduction toward a higher proportion of fat mass compared with muscle tissue.
Why Lean Mass Preservation Is Important
GLP-1–based medications help you lose fat, but you may also lose muscle. That shift can affect strength, metabolic health, and long-term weight maintenance.
By testing trevogrumab as an add-on therapy, researchers aimed to determine whether you can improve fat loss while limiting muscle reduction, without compromising tolerability. Ongoing studies continue to evaluate durability and safety outcomes.
Potential Applications Being Studied
Researchers are testing trevogrumab in settings where you risk losing muscle, especially during pharmacologic weight loss and the weight-maintenance phase.
Current investigations include:
- Preserving lean tissue during fat mass reduction with GLP-1–based drugs
- Use in sarcopenia and other muscle-wasting disorders
- Metabolic disease contexts, including obesity
- Evaluation in triplet therapy or other triplet combination strategies
You cannot access it outside controlled clinical trials.
Why Protecting Lean Body Mass Matters
When you preserve muscle mass during weight loss, you support a higher resting metabolic rate and better glucose control. Stronger lean body mass improves physical function and daily performance.
Limiting lean mass loss also supports long-term weight stability and may reduce cardiovascular risk. As obesity treatments advance, clinicians now prioritize lean mass preservation to improve the quality—not just the amount—of weight reduction.
Is Trevogrumab FDA Authorized?
No. You cannot access trevogrumab as an approved therapy because the FDA has not authorized it.
Regeneron Pharmaceuticals is developing it with its VelocImmune platform, and studies continue in controlled trials, including combinations with semaglutide from Novo Nordisk.
- Status: Investigational
- Availability: Clinical trials only
Where Trevogrumab Research Is Heading
Research is shifting your focus from simple scale reduction to improving body composition.
Instead of targeting total pounds lost, investigators now examine how therapies can reduce fat while protecting lean tissue. Ongoing trials will clarify whether trevogrumab fits into future combination strategies designed to help you lose fat without sacrificing muscle mass.
Frequently Asked Questions About Trevogrumab
What conditions is trevogrumab being studied for?
Researchers are testing trevogrumab for muscle preservation and body composition support during weight loss treatment.
You may see it studied alongside GLP‑1 medications to help maintain lean mass while reducing fat. It remains an investigational therapy.
Can trevogrumab increase muscle size?
Trevogrumab blocks myostatin, a protein that limits muscle growth.
This mechanism may support greater muscle development, but clinical trials are still evaluating how much muscle gain actually occurs in patients.
Can you buy trevogrumab right now?
You cannot purchase trevogrumab.
It is an experimental monoclonal antibody available only through supervised clinical trials.
What do we know about its safety profile?
Clinical trials continue to assess safety, tolerability, and treatment‑emergent adverse events.
Reported side effects may include nausea, headache, and muscle spasms. Researchers also monitor serious events such as cardiac arrest, along with treatment discontinuation rates due to adverse events.
Frequently Asked Questions
How does trevogrumab work to protect muscle tissue?
Trevogrumab is a human monoclonal antibody that targets myostatin (also called GDF‑8), a protein that limits muscle growth. When you block myostatin, you reduce one of the body’s natural signals that slows muscle development.
By neutralizing this signal, trevogrumab aims to help you maintain or increase lean muscle mass, especially during periods of weight loss or physical inactivity.
Researchers have tested it alongside GLP‑1 medications such as semaglutide to see whether it can help you lose fat while preserving muscle.
What side effects have researchers observed so far?
Trevogrumab remains under clinical investigation, so safety data are still developing.
Because it is an injectable biologic, you may experience:
- Injection site reactions
- Mild flu‑like symptoms
- Headache or fatigue
Serious adverse events have not been widely reported in early‑phase obesity and muscle studies, but long‑term risks remain under evaluation. You should expect ongoing monitoring in clinical settings.
When might trevogrumab and garetosmab become available?
Trevogrumab and the related antibody garetosmab are not approved for general medical use.
Both agents remain in clinical development. Regulatory approval depends on successful completion of larger trials that confirm safety, effectiveness, and long‑term outcomes.
There is no confirmed release date for routine clinical prescribing.
What have recent trevogrumab trials shown?
Recent Phase 2 research, including the COURAGE trial, evaluated trevogrumab in people with obesity.
Key findings include:
| Study Focus | Reported Outcome |
|---|---|
| Trevogrumab alone | Trends toward preserving lean mass |
| Combined with semaglutide | Greater fat loss with better muscle retention compared to semaglutide alone |
| Metabolic markers | Early signals of possible lipid profile improvement |
These findings suggest that blocking myostatin may help you maintain muscle while reducing body fat, but larger studies must confirm these results.
What is trevogrumab’s current trial phase and expected goals?
Trevogrumab has reached Phase 2 clinical trials for conditions such as sarcopenia, obesity‑related muscle loss, and disuse atrophy.
Ongoing and future studies aim to determine:
- Whether lean mass preservation translates into better strength and function
- Long‑term safety with repeated dosing
- Optimal use alongside incretin‑based weight‑loss therapies
You should expect additional mid‑ to late‑stage trials before regulators consider approval.
What could trevogrumab treatment cost in the future?
There is no official pricing because the drug is not approved.
However, monoclonal antibody therapies often cost thousands to tens of thousands of dollars per year, depending on dosing and indication. Final pricing will depend on manufacturing costs, treatment duration, insurance coverage, and regulatory decisions.
At this stage, any cost estimate remains speculative.